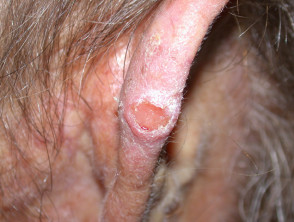
Cemiplimab disponibile in Italia per due nuove indicazioni: carcinoma basocellulare e tumore al polmone non a piccole cellule - AboutPharma

Overview of Libtayo, First New FDA-Approved Drug for Metastatic Cutaneous Squamous Cell Carcinoma (CSCC) | Everyday Health

Overview of Libtayo, First New FDA-Approved Drug for Metastatic Cutaneous Squamous Cell Carcinoma (CSCC) | Everyday Health